Journal of Diabetes Management and Metabolism
Research Article
Allantoin and Micronutrients Restores Renal Dysfunction in Streptozotocin Induced Diabetic Rats.
Ganesha KR*, Razdan R and Minaz N
Department of Pharmacology, Al-Ameen College of Pharmacy, India
*Corresponding author: Ganesha KR, Department of Pharmacology, Al-Ameen College of Pharmacy, India, Tel: +917760653948; Email: krganesh87222g@gmail.com
Citation: Ganesha KR, Razdan R and Minaz N (2019) Allantoin and Micronutrients Restores Renal Dysfunction in Streptozotocin Induced Diabetic Rats. J Diabetes Metab Manag: JDMM-10004
Received date: 10 June, 2019; Accepted date: 24 June, 2019; Published date: 05 August, 2019
Abstract
Objectives: Diabetic nephropathy is the major complication in diabetic patients. The current available treatment options for diabetic nephropathy do not affect the underlying pathology and slow the progression of the disease. The many experimental data suggest that individual component of micronutritional supplementsand allantoin has shown the protective effect in diabetic nephropathy. So with the aim to evaluate the combination of allantoin and micronutrientsin diabetes induced nephropathy.
Methodology: Diabetes was induced in male Wistar rats by administrating streptozotocin (STZ) (52mg/kg, i.p). Diabetic rats were treated with allantoin (100 and 200 mg/kg), nutritional supplement, combination of allantoin (100 mg/kg) + nutritional supplement and allantoin (200 mg/kg) + nutritional supplement for 6 weeks. After the six weeks of treatment body weight, renal function parameters and kidney index were measured. Antioxidant enzymes (SOD) and lipid peroxide levels were determinedin the kidney homogenateand histopathology of kidney was carried out.
Results: The combination therapy of allantoin and micro nutritional supplements weresignificantly elevated the body weight, significantly reduced serum glucose level, glycosylated haemoglobin, and significantly improved renal function parameters. In kidney homogenate, the treatment with combination therapy increases the antioxidant enzyme level and reduces the lipid peroxide product compared to diabetic rats.
Conclusion: The combination therapy of allantoin and micronutritional supplements has improved glycemic control in diabetic rats. Hence, treatment with combination of drugs may be a way to offset the early pathophysiologic damage resulting from hyperglycemia which can be attributed towards its nephroprotective effects in rats.
Keywords: Allantoin; Diabetic nephropathy; Nutritional supplements (Benfotiamine + gamma linolenic acid + Alpha lipoic acid + L-methyl folate + methylcobalamin + Pyridoxal-5-phosphate).
Introduction
Diabetes mellitus (DM) is currently a major health problem all over the world and is a chronic metabolic disorder resulting from a variable interaction of hereditary and environmental factors. Diabetic nephropathy is an important complication of both types of DM. Diabetic nephropathy is associated with the increase mortality in diabetic patients and this result in the end stage renal disease in most of the countries. DiabeticNephropathy is reported to develop in 30-40% of patients with diabetes and has become a leading cause of end stage renal failure worldwide.People with diabetes can develop kidney damage by increasing the pressure in the delicate filtering system of the kidney. Diabetic nephropathy also appears to be more common in people who have problems controlling their blood glucose, as well as those with high levels of blood fat and blood pressure and those who are overweight [1,2].Treatment first involves bringing blood glucose levels within the normal range. In this study the combination of drugs is used to treat diabetic nephropathy.
Allantoin exhibits anti-diabetic effects by modulating antioxidant activities, lipid profile and by promoting release of glucagon like peptide (GLP-1), thereby improving the function of β-cells maintaining normal insulin and glucose level. Allantoin is also found to increase nitric oxide (NO) level [3]. The micronutrients (benfotiamine, gammalinolenic acid, alpha lipoic acid, L-methylfolate, methylcobalamine, pyridoxal-5-phosphate) have shown essential roles in diabetic patients.
In experimental diabetes, thiamine and benfotiamine (a synthetic S-acyl derivative of thiamine) supplement prevented tissue accumulation and increased urinary excretion of protein glycation, oxidation and nitration adducts.Alpha-lipoicacid supplement is a unique biological antioxidant because it is soluble in both water and lipids.This allows it to neutralize free radicals just about everywhere in the body, inside and outside the cells.Due to its unique sulfur-containing structure, alpha-lipoic acid can scavenge several types of free radicals. Gamma linolenic acid produced in the body from linolenic acid an essential fatty acid of omega-6 series by the enzyme delta-6-desaturase. GLA forms di homo-gamma linolenic acid which can be converted to prostaglandin E1. PGE1 has anti-inflammatory, antiplatelet and vasodilatory properties. In patients with diabetes decreased production of PGE1 has been reported. L-methyl folate is a biologically active form of folate used at the cellular level for DNA reproduction. It maintains the appropriate levels of NO production cause vasodilatation and are important in protecting an organ from ischemic damage and ischemic pain.Methylcobalamin Methyl cobalamin is a form of vitamin B12. It also increases use of glucose and fatty acids for ATP production. Pyridoxal-5-phosphate is an active form of vitamin B6, is a coenzyme in a variety of enzymatic reactions. P5P helps to balance sodium and potassium levels by regulating the electrical functioning of nerves, heart and musculoskeletal system. P5P also helps body to build and break down amino acids and to change amino acids from one to another [4-9]. All these above mentioned micronutrients shown beneficial effect in diabetic nephropathy. So considering all, we evaluated protective effect of allantoin and micronutrients in diabetic nephropathy.
Methods and Materials
Animals: Male albino Wisterrats (weight, 220 to 250 g) were obtained from Central Animal House, Al-Ameen College of Pharmacy. They were maintained in standard laboratory conditions (temperature, 22 ± 2°C and relative humidity, 55 to 60%). They were fed with standard pellet diet and water ad libitum. The study protocols were approved by Ethics Committee (AACP/IAEC/feb2018/02) and the experiments were conducted in accordance with guidelines set by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA).
Drugs and pharmaceuticals: Allantoin was obtained from Himedia Laboratories Pvt. Ltd (Karnataka, India). The streptozotocin was obtained from M.P biomedicals and all the diagnostic kits were purchased from Autospan. The Maltida AF capsules were used for the micronutritional supplements from Lupin, Ltd.
Experimental design
Induction of diabetic nephropathy: Type 1 diabetes was induced by streptozotocin (52 mg/kg. of body weight i.p. in 100 mM Sodium citrate in ice cold buffer, pH 4.5). The elevated serum glucose was determined after 3 days of streptozotocin administration and those rats with fasting glucose levels >250 mg/dL were served as diabetic rats.The rats were divided into following groups. Group 1 was the normal control; Group 2, Diabetic control; Groups 3 and 4, DC + low-dose and high-dose allantoin (AL, 100 mg/kg and 200 mg/kg, p.o. to 6 weeks respectively); Group 5, DC + micronutritional supplements (MN, p.o. to 6 weeks); Groups 6 and 7, DC+MN+AL (100 mg/kg and 200 mg/kg p.o. to 6 weeks respectively). We selected dose of allantoin according to the previous studies where showed these doses to be effective. The dose of micronutrients was selected based on the conversion of human dose into the animal dose.
Biochemical assays: After 6 weeks of treatment the blood was withdrawn from retro-orbital plexus. Blood was collected in EDTA storage tubes and the samples were used to analyze the serum blood glucose, glycosylated hemoglobin, blood urea nitrogen levels. Then the animals were kept for 24 hours in metabolic cage for urine collection. Urine volume was measured and further it was used for the analysis of urine creatinine, and albumin levels [10]. The urine and blood parameters were evaluated using a semi-auto-analyzer following suitable methods by commercially available kits.After the biochemical assays the animals were sacrificed by overdose of anesthesia, and the kidneys were dissected out. Then the right kidneys were (patted dry, weighed) minced then homogenized for estimation of antioxidant enzymes.
Estimation of super oxide dismutase: Tissue homogenate preparation: 2% w/v of the tissue was taken and 10%w/v of buffer was added and homogenate was prepared. 0.1 ml of sample was mixed with 0.1 ml of EDTA (1 × 10-1 M), 0.5 ml of carbonate buffer (pH 9.7) and 1 ml epinephrine (3 ×10-3 M). The optical density of formed adenochrome was measured at 480 nm for 3 minutes at an interval of 30 seconds. The results were expressed as unit per mg protein [11].
Estimation of lipid peroxidation: Tissue extracts were mixed separately with 1 ml TCA (20%), 2 ml TBA (0.67%) and heated for 1 h at100°C. After cooling, the precipitate was removed by centrifugation. The absorbance of each sample was measured at 535 nm using a blank containing all the reagents except the sample. As 99% TBARS are malondialdehyde (MDA), so TBARS concentrations of the samples were calculated using the extinction coefficient of MDA, which is 1.56 × 105 M-1 cm-1 and were expressed as μmol of malondialdehyde per mg protein [11].
Histopathological Studies: Then, the left kidneys were stored in 10% buffered formalin solution (formaldehyde, 100 mL; sodium phosphate monobasic, 4 g; sodium phosphate dibasic, 6.5 g; and water, 900 mL) and subjected to further processing for histopathological evaluation.
Results
These results were better improvements in renal dysfunction in diabetic nephropathy in diabetic rat model.
Effect of allantoin and nutritional supplement on the percentage change in body weight in STZ induced diabetic rats: A Significant percentage reduction in body weight of diabetic rat was observed when compared with normal rats. The diabetic rats treated with allantoin (100 and 200 mg/kg), micronutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement, significantly reduced the % change in body weight of diabetic rats (Figure 1).
Effect of allantoin and nutritional supplement on serum parameters
The % change serum glucose level in diabetic rats was significantly higher than normal rats: The serum glucose levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement, were significantly reduced as compared to diabetic control rats (Figure 2).
The %GHb of diabetic rats was significantly higher than normal rats: The %GHb of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement was significantly reduced as compared to diabetic control rats (Figure 2).
Effect of allantoin and nutritional supplement on urine parameters
In the diabetic rats, urine volume was significantly increased when compared to the normal rats:In the diabetic rat’s treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement, a significantly lower urine volume was observed when compared to the diabetic rats.
A urinary albumin level in STZ diabetic rats was significantly higher than normal rats: The urinary albumin levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) + nutritional supplement were significantly reduced as compared to diabetic control rats.
Urinary creatinine levels in STZ diabetic rats were significantly lower than normal rats: The urinary creatinine levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) + nutritional supplement and allantoin (200 mg/kg) + nutritional supplement were significantly increased as compared to diabetic control rats.
Creatinine clearance levels in STZ diabetic rats were significantly lower than normal rats: The creatinine clearance levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) + nutritional supplement were significantly increased as compared to diabetic control rats (Figure 3,4), Histopathological evaluation (Figure 5).
Effect of allantoin and nutritional supplement on kidney index in diabetic rats: Kidney index levels in diabetic rats were significantly higher than normal rats. The kidney index levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement were significantly reduced as compared to diabetic rats (Table 1).
Effect of allantoin and nutritional supplement on serum BUN level in diabetic rats: Serum BUN levels in STZ diabetic rats were significantly higher than normal rats. The serum BUN levels of diabetic treated with allantoin (100 and 200 mg/kg), nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement were significantly decreased as compared to diabetic control rats (Table 1).
Effect of allantoin and nutritional supplement on super dismutase (SOD) and lipid peroxidation levels in kidney homogenate of diabetic rats: SODlevels in diabetic rats were significantly lower than normal rats. The SODlevels of diabetic treated with allantoin (100 and 200 mg/kg),nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement was significantly increased as compared to diabetic rats.
Lipid peroxidation level in diabetic rats was significantly higher than normal rats. The lipid peroxidation level of diabetic treated with allantoin (100 and 200 mg/kg),nutritional supplement, allantoin (100 mg/kg) +nutritional supplement and allantoin (200 mg/kg) +nutritional supplement was significantly decreased as compared to diabetic rats.
Discussion
In the present study we used a diabetogenic agent streptozotocin to induce experimental DN. As it selectively destroys the islets of Langerhans, chronic hyperglycemia was observed in diabetic rat throughout our study. Treatment with combination therapy of allantoin and micro nutritional supplements were reducing blood glucose level. Our results are in accordance with study in which allantoin and micronutritional supplements with antihyperglycemic effect. Allantoin exhibits anti-diabetic effects by modulating antioxidant activities, lipid profile and by promoting release of glucagon like peptide (GLP-1), thereby improving the function of β-cells maintaining normal insulin and glucose level. Allantoin is also found to increase nitric oxide (NO) level. Our results are in accordance with the study of the combination therapy of allantoin and micro nutritional supplements thus confirming the anti-hyperglycemic activity [12].
In Type 1 diabetes mellitus an elevation in the glucose level is directly proportional to the rate of glycosylation of hemoglobin causing the formation of GHb (glycosylated hemoglobin) over previous four weeks to three months. Marked increase in percentage of GHb has been reported in previous studies in diabetic rats13.GHb formation has been implicated in various diabetic micro vascular complications like neuropathy, nephropathy, retinopathy etc. Previous studies revealed the role of glycosylation leading to the formation of oxygen-derived free radicals in diabetes mellitus and its level can be considered as one of the important marker of oxidative stress[13]. Therefore, in the present study we have measured the level of GHb as a marker of diabetes induced oxidative stress. Our results were showed that the level of GHb in diabetic rats was higher as compare to the normal rats. The combination of allantoin and nutritional supplements significantly reduced % glycosylated hemoglobin this protective effect could be due to its anti-hyperglycemic effect.
The proteinuria that develops after induction of diabetes is mainly due to an increased excretion of an array of low molecular weight proteins where albumin comprises a relatively low proportion (5-20%) of total urinary protein. The mechanism for increased urinary excretion of low molecular weight proteins has been considered generally due to decreased tubular reabsorption of filtered plasma proteins. Increased glomerular permeability causes plasma proteins, to enter the ultra-filtrate. The damage to glomerular membrane and loss of negative charge from the filtration slit resulting in increased filtration of albumin and damage to the tubules causing increased albuminuria.
Creatinine clearance an indicator of decrease in glomerular filtration rate is due to glomerular hyperfilteration increased glomerular capillary pressure and leaky glomerular capillaries, as well as impaired tubuloglomeruli. As the glomerular damage progresses, the thickened capillary basement membrane ultimately decreases the rate of diffusion and results in a decrease in GFR. In the present study, there was an elevation in urinary albumin and reduction in urinary creatinine, creatinine clearance in the diabetic rats indicating renal damage [13]. Treatment of diabetic rats with allantoin and nutritional supplements effectively reduced the level of urinary albumin and elevation in urinary creatinine, creatinine clearance levels as compared to the diabetic rats.
Blood urea nitrogen is the marker of renal function. The BUN that rises after induction of diabetes is mainly due to increased amount of urea nitrogen in blood. Increasing serum BUN level has been reported as waste products of metabolism following kidney injury and they have known biomarkers of kidney degeneration. In this study, the elevation of plasma BUN with hyperglycaemia can be proposed as indicator for renal dysfunction [14,15].
The combination of allantoin and nutritional supplements significantly reduced BUN level due to its anti-hyperglycemic effect.
Increase in the weight of kidney (hypertrophy) in proportion to the body weight was observed in diabetic rats. Local alterations in the production of one or more growth factors such as over expression of transforming growth factor - beta 1 in the kidney especially in proximal convoluted tubules cells and glomerular mesangial cells are proposed in the development of renal hypertrophy. An increase in the rate of protein synthesis or decrease in the degradation of renal extracellular components might also lead to renal hypertrophy [16]. The combination therapy of allantoin and micronutrients were reduced kidney index thus demonstrating reversal of kidney hypertrophy in diabetic rats.
Oxidative stress ensues due to the hyperactivity of polyol pathway induced by hyperglycemia and acts as major promoter of diabetic complications including DN [17]. It is well accepted that, the high oxidative stress in diabetics considerably contributes to the complication of this disease [18] and excessive production of free radicals is an observed phenomenon in association with diabetic complications. Various metabolic pathways like AGE pathway, polyol pathway, PKC pathway and hexosamine pathway directly or indirectly alter the redox capacity of the cell and increase the production of ROS. STZ exerts its diabetogenic action via increased production of ROS and inhibition of anti-oxidative enzyme (SOD) that scavenges ROS [19]. Increased concentration of lipid peroxidation (LPO) markers viz., TBARS were observed in kidney of experimental animals. This increased level of lipid peroxidation could be associated to increase in free radicals' generation in diabetes caused primarily due to high blood glucose levels, which upon autoxidation generates free radicals [20].
The great antioxidant potential of allantoin and micronutrients has been proven, these antioxidants has gained an important role in the treatment for diseases that present high production of free radicals, especially metabolic disorder such as diabetes [21-27]. Treatment of diabetic rats with allantoin and nutritional supplements effectively reduced lipid peroxidation levels and increased SOD levels. Histopathological examinations of kidney sections of H&E stained diabetic rats were showed positive for focal mesangial sclerosis, focal aggregates of inflammatory cells. The combination therapy of allantoin and micronutrients were significantly reduced the aforementioned alterations, thus demonstrating protective role in renal damage.
Conclusion
Our finding suggests the positive effects of combination therapy on both functional and structural abnormalities in diabetic induced nephropathy by exerting anti-hyperglycemic and antioxidant effect. Hence, the combination therapy may be a way to offset early pathophysiologic damage resulting from hyperglycemia which can be attributed towards its nephron protective effects in rats.
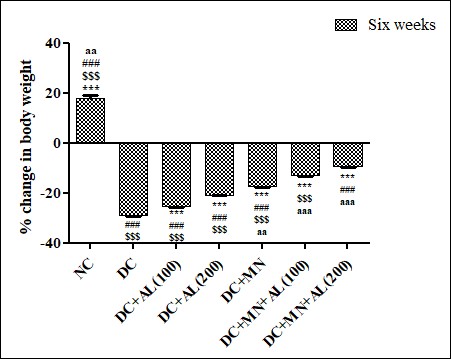
Figure 1: Effect of allantoin and nutritional supplement on the percentage change in body weight in STZ induced diabetic rats. Values are represented as mean ± SEM (n=6). One Way ANOVA followed by Tukey’s Multiple Comparison Test. *p<0.05, **p<0.01, ***p<0.001 Vs Diabetic control group, #p<0.05, ##p<0.01, ###p<0.001 Vs Diabetic + Allantoin (100 mg/kg) + Micronutrients (DC+AL100+MN), $p<0.05, $$p<0.01, $$$ p<0.001 Vs Diabetic + Allantoin (200 mg/kg) + Micronutrients (DC+ AL200+MN). aap<0.01, aaap<0.001 for sixth week group Vs third week group. NC: Normal control, DC: Diabetic control, MN: Micronutrients, AL: Allantoin.
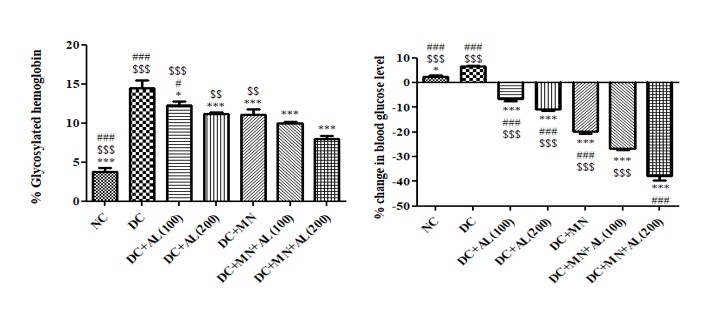
Figure 2: Effect of allantoin and nutritional supplement on % change in serum glucose level and % glycosylated hemoglobin in diabetic rats. Values are represented as mean ± SEM (n=6). One Way ANOVA followed by Tukey’s Multiple Comparison Test. *p<0.05, **p<0.01, ***p<0.001 Vs Diabetic control group, #p<0.05, ##p<0.01, ###p<0.001 Vs Diabetic + Allantoin (100 mg/kg) + Micronutrients (DC+AL100+MN), $p<0.05, $$p<0.01, $$$p<0.001 Vs Diabetic + Allantoin (200 mg/kg) + Micronutrients (DC+ AL200+MN).NC:Normal control, DC:Diabetic control, MN: Micronutrients, AL: Allantoin.
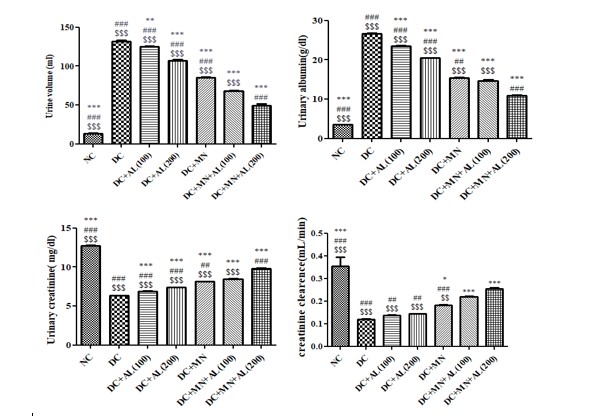
Figure 3: Effect of allantoin and nutritional supplement on urine volume, urinary albumin, creatinine, and creatinine clearance levels in diabetic rats. Values are represented as mean ± SEM (n=6). One Way ANOVA followed by Tukey’s Multiple Comparison Test. *p<0.05, **p<0.01, ***p<0.001 Vs Diabetic control group, #p<0.05, ##p<0.01, ###p<0.001 Vs Diabetic+Allantoin (100 mg/kg) + micronutrients (DC+AL100+MN), $p<0.05, $$ p<0.01, $$$ p<0.001 Vs Diabetic+Allantoin (200 mg/kg) + Micronutrients (DC+AL200+MN). NC: Normal control, DC:Diabetic control, MN: Micronutrients, AL: Allantoin.
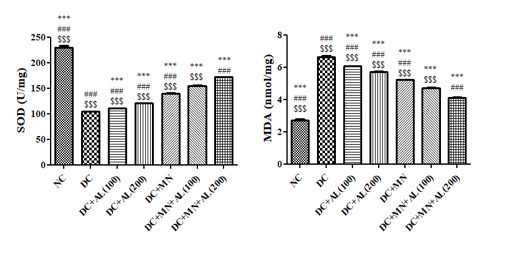
Figure 4: Effect of allantoin and nutritional supplement on SOD and MDA levels in kidney homogenate of diabetic rats. Values are represented as mean ± SEM (n=6). One Way ANOVA followed by Tukey’s Multiple Comparison Test. *p<0.05, **p<0.01, ***p<0.001 Vs Diabetic control group, #p<0.05, ##p<0.01, ###p<0.001 Vs Diabetic + Allantoin (100 mg/kg) + Micronutrients (DC+AL100+MN), $p<0.05, $$p<0.01, $$$p<0.001 Vs Diabetic + Allantoin (200 mg/kg) + Micronutrients (DC+ AL200+MN). NC: Normal control, DC: Diabetic control, MN: Micronutrients, AL: Allantoin.
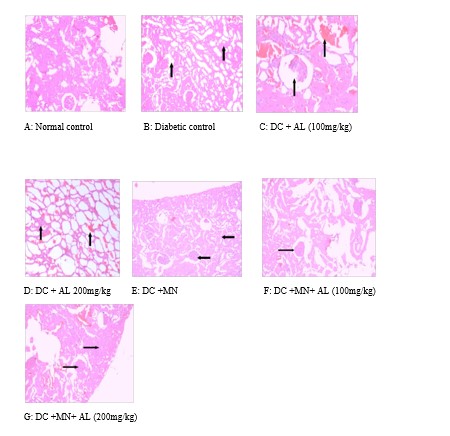
Figure 5: Effect of combination therapy on histopathology of kidney of normal rats, STZ-induced diabetic rats (DM), treatment groups. (A) Normal control group showing glomerulus and tubules. No area of hemorrhage or necrosis seen. (B) Diabetic control group showing focal mesangial sclerosis, focal aggregates of inflammatory cells. (C) DC+AL (100mg/kg) group showing poorly reducing the mesangial sclerosis and the fatty and hyaline change in tubules. Hemorrhage or necrosis has seen in tissue. (D) DC+AL (200mg/kg) group showing reducing the mesangial sclerosis and reduce the fatty and hyaline change in tubules. Hemorrhage or necrosis has seen in tissue. (E) DC+MN group showing reducing the mesangial sclerosis and reduce the fatty and hyaline change in tubules. No hemorrhage or necrosis has seen in tissue. (F) DC+MN+AL (100mg/kg) and (G) DC+MN+AL (200mg/kg) group showing decreased in mesangial matrix and tubules with degenerative changes (H&E staining (100X).
|
Sl.No |
Groups |
Kidney index (mg/g) |
Serum BUN levels(mg/dl) |
|
1 |
Normal control |
4.35 ± 0.42*** ###$$$ |
18.51 ± 0.054*** ###$$$ |
|
2 |
Diabetic control |
8.35 ± 0.55### $$$ |
85.15 ± 0.086### $$$ |
|
3 |
Diabetic+ Allantoin (100 mg/kg) |
8.09 ± 0.18*** ### $$$ |
80.40 ± 0.276*** ###$$$ |
|
4 |
Diabetic +Allantoin (200 mg/kg) |
7.84 ± 0.29*** ### $$$ |
69.03 ± 0.640*** ###$$$ |
|
5 |
Diabetic+ nutritional supplement |
7.34 ± 0.52*** ### $$$ |
62.24 ± 0.764*** ### $$$ |
|
6 |
Diabetic+ Allantoin (100 mg/kg) +nutritional supplement |
7.04 ± 0.23*** $$$ |
56.16 ± 0.322*** $$$ |
|
7
|
Diabetic+Allantoin(200 mg/kg) +nutritional supplement |
6.34 ± 0.18*** ### |
48.55 ± 0.482*** ##
|
|
Note: Values are represented as mean ± SEM (n=6). One Way ANOVA followed by Tukey’s Multiple Comparison Test. *p<0.05, **p<0.01, ***p<0.001 Vs Diabetic control group, #p<0.05, ##p<0.01, ###p<0.001 Vs Diabetic + Allantoin (100 mg/kg) + Micronutrients (DC+AL100+MN), $p<0.05, $$p<0.01, $$$p<0.001 Vs Diabetic + Allantoin (200 mg/kg) + Micronutrients (DC+ AL200+MN). |
|||
Table 1: Effect of allantoin and nutritional supplement on kidney index and serum BUN level in diabetic rats.
Citation: Ganesha KR, Razdan R and Minaz N (2019) Allantoin and Micronutrients Restores Renal Dysfunction in Streptozotocin Induced Diabetic Rats. J Diabetes Metab Manag: JDMM-10004