Emerging Infectious Diseases and Diagnosis Journal
Case Report
Overlap of Toxic Epidermal Necrolysis and Cutaneous Drug Induced Lupus-A Case Report
Khan K and Qureshi S*
Department of medicine and allied, Northwest General Hospital and Research Centre, Peshawar, Pakistan
*Corresponding author: Sameed Qureshi, Department of medicine and allied, Northwest General Hospital and Research Centre, Peshawar, Pakistan, Tel: +923349057484; Email: sameedqureshi92@gmail.com
Citation: Khan K and Qureshi S (2019) Overlap of Toxic Epidermal Necrolysis and Cutaneous Drug Induced Lupus-A Case Report. Emerg Infect Dis Diag J: EIDDJ-100005
Received date: 19 September, 2019; Accepted date: 26 September, 2019; Published date: 07 October, 2019
Abstract
A 34-year-old lady presented with generalized rash for the last three weeks and use of polypharmacy. The clinic pathological correlation was most consistent with the diagnosis of toxic epidermal necrolysis. Labs were remarkable for thrombocytopenia, acute kidney injury, positive anti-nucleosome, anti dsDNA and anti-histones antibodies. Our patient improved with conservative treatment and withdrawal of drugs. This case highlights a challenging clinical differential diagnosis of overlap between TEN and drug induced lupus erythematosus.
Keywords: Drug induced; SLE; Toxic epidermal necrolysis
Introduction
Toxic epidermal necrolysis(TEN) is a life-threatening dermatological disorder characterized by diffuse erythema, bullous detachment of epidermis and mucous membranes, necrosis resulting in exfoliation of skin which can be complicated by sepsis or even death [1].
Differentiating TEN and rash of SLE can be difficult, as both share clinical and histological findings such as diffuse desquamation, mucosal erosions, and keratinocyte necrosis. Fatigue, myalgias, and polyarthritis may be part of the clinical symptoms. A positive ANA, anti dsDNA and a prolonged disease course favours SLE, while an identifiable trigger such as drugs and more acute onset favours Ten.
Our patient had a history of polypharmacy with ingestion of multiple antibiotics, NSAIDs and steroids among other drugs. She developed a diffuse rash which was clinically indistinguishable between TEN or cutaneous lupus erythematosus. Based on the earlier history with multiple admissions and disease manifestations, there was an overlap between clinical entities. Conservative management with fluids, cessation of drugs, good oral intake and nursing care was done. Here we report good outcome of using this strategy for such patients.
Case Report
This thirty-four-year-old-lady from Afghanistan was presented to Northwest General Hospital in the month of May, 2019 with a generalized rash all over the body for the last three weeks. The patient has been admitted prior three times in the last two months for repeated polypharmacy including multiple antibiotics and NSAIDs use and previous admission with acute pancreatitis and pancytopenia. The patient was discharged two months back with normal count and resolved pancreatitis. The patient was fine a month back when she developed generalized body aches, the patient would go visit multiple quacks and doctors where she would receive multiple antibiotic, painkillers and steroids. The patient was advised to be screened for autoimmune disease which she did not do and received multiple drugs whenever she would get aches and undocumented fever. She developed a rash. It rapidly progressed into a generalized erythematous rash with facial puffiness and conjunctivitis. The patient was toxic, febrile, had polyarthralgia. In the next two weeks, her condition deteriorated with extension of skin lesions, rise in body temperature and pulse rate.
On arrival the patient was hypotensive with blood pressure 100/60 and febrile with temperature of 100F. Dermatological examination had generalized skin involvement, affecting >95% body surface area sparing only the antecubital and popliteal fossa. The skin was markedly erythematous, oedematous, tender and peeling off as shown in Figure 1. Nikolsky's sign was positive and few flaccid bullae were seen in the dependent areas. In addition, she had genital mucosal sloughing. Rest of the systemic examination was unremarkable.
A clinical diagnosis of toxic epidermal necrolysis was made and the patient received good nursing care.Initially, laboratory investigations revealed mild leukocytosis, thrombocytopenia, sepsis, acute renal injury, raised bilirubin with alkaline phosphatase and low albumin level. Serology for HIV, hepatitis B and C, and hemocultures were negative. INR 2.0. Investigations are shown in Table 1.
She was started on intravenous crystalloids with ringer lactate, antiemetic, vitamin K, folic acid and strict intake output record was done. She was encouraged to take orally and high protein diet was advised. Ultrasound abdomen and pelvis was normal. She was given a bed bath daily. Cutaneous mucosal ulceration was washed with saline and physio gel ointment was applied. Pus swab grew methicillin resistant staphylococcus aureus. All her previous medications including antibiotics, steroids, non-steroidalanti-inflammatory drugs (NSAIDs) were stopped. She was started on levofloxacin when she spiked fever. The underlying medical condition was sought for multiorgan involvement. Autoimmune screening was positive for ANA, dsDNA antibodies, histones, nucleosome antibodies were strongly positive confirming the diagnosis of background systemic lupus erythematosus. She was prescribed hydroxychloroquine 200 mg q12 hours. Significant improvement was seen within 4 days, with stabilization of vital parameters, absence of fresh bullae, reduction of erythema and exudation from the skin. She improved over the next week with subsidence of lesions over the trunk and extremities. Body flexures and genitalia were last to recover. Hypopigmentation on the face, trunks, body and back is resolving. The patient and the attendants have been counseled in detail regarding the unnecessary use of antibiotics or any other drugs unless prescribed in the future.
Discussion
Toxic epidermal necrolysis is a life threatening condition which results in erythematous blisters and eruptions caused mostly by the idiosyncratic drug reactions [2]. However, it can also be caused in sepsis, vaccinations, graft vs host disease and SLE [3]. SLE is a chronic inflammatory disease of unknown cause that can affect virtually every organ. Patients may present with a wide array of symptoms, signs, and laboratory findings and have a variable prognosis that depends upon the disease severity and type of organ involvement. The skin being the most commonly involved [4]. Clinical picture in both conditions may be unspecific (fever, generalized body aches and lethargy) and preceded by cutaneous manifestations. The early skin lesions include erythematous and livid macules which is followed by hemorrhagic erosions and erythema, epidermal detachment, which present as blisters and areas of denuded skin. The diagnosis relies on clinical symptoms and on histological examination [5]. Diagnosis of TEN is based on both clinical and histologic findings. Early TEN can resemble non-specific drug reactions, so clinicians should maintain a high index of suspicion for TEN. The Nikolsky sign is also helpful diagnostic signs found in patients with TEN [6]. Differential diagnosis includes TEN, cutaneous SLE, erythema multiform, viral exanthems, and other drug rashes. TEN can usually be differentiated clinically as the disorder evolves and is characterized by significant pain and skin sloughing [6]. SLE on the other hand follows the criteria set developed by the American College of Rheumatology which includes person having SLE if any 4 out of 11 symptoms were present simultaneously or serially on two separate occasions. The criteria include malar rash, discoid rash, serositis or pericarditis, oral ulcers, arthritis, arthritis, photosensitivity, hematological disorder, renal involvement, ANA, immunologic disorder including positive anti-Smith, anti-ds DNA, antiphospholipid antibody or false positive serological test of syphilis and neurological disorders [7].
In our case differential diagnosis of drug-induced TEN and other autoimmune conditions was considered. However, there was evidence of high risk drugs in our case. Several case reports have been published in the literature. A study conducted by had 1366 patients with SJS/TEN, 17 with a sufficiently documented history of SLE and representative histological material could be identified. Eight of these showed clinically and histopathologically characteristic features interfering with the diagnosis of TEN, four of these had SJS/TEN with a preceding SLE exacerbation and four as TEN?like SLE [8].
Another paper by Brandon.T.et all had acute systemic lupus erythematosus presenting as toxic epidermal necrolysis with less than 25 reported cases [9]. As more cases are reported, larger studies may improve our understanding of the risk factors for this. At present, no treatment modality has been established as a standard for these patients with overlap of TEN and SLE. Differentiating these two entities has practical and important implications for patients, treatment, and counseling. We present a rare case of TEN and SLE that required treatment with conservative approach and nursing care. This case highlights a challenging clinicopathologic differential diagnosis important for dermatologists and dermatopathologists to be aware of.
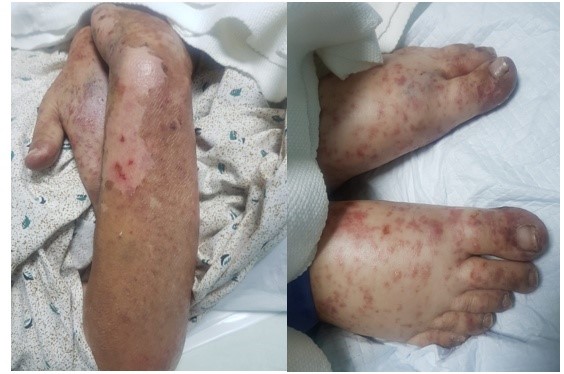
Figure 1:The skin was markedly erythematous, oedematous, tender and peeling off.
|
Laboratory Investigations |
Value |
Normal Range* |
|
Hemoglobin |
10.42 |
13-18 g/dL |
|
Red blood cell count |
3.5 |
4.5-5.5 × 10 ^ 12/L |
|
Hematocrit |
29.9 |
40-54% |
|
MCV |
85.6 |
83-101 fl |
|
Platelets |
20.93 |
150-450 × 10 ^ 9/L |
|
White cell count |
14.22 |
4-11 × 10 ^ 9/L |
|
Differential Leukocytes |
||
|
Neutrophils |
10.95 |
1.65-8.25 × 10 ^ 9/L |
|
Lymphocytes |
1.56 |
0.8-4.95 × 10 ^ 9/L |
|
Eosinophils |
0.28 |
0.04-0.66 × 10 ^ 9/L |
|
Monocytes |
1.42 |
0.24-1.1 × 10 ^ 9/L |
|
Other tests |
||
|
Creatinine |
2.67 |
0.2-1.2 mg/dl |
|
Random glucose |
96 |
110-165 mg/dl |
|
PT |
20 |
10.0 sec |
|
ALT |
31 |
10-40 U/L |
|
Malarial Parasite |
Not seen |
|
|
CRP |
10.2 |
7.51 mg/dl |
|
Calcium |
8.8 |
8.5-10.5 mg/dl |
|
HCV Antibody |
Non-Reactive |
- |
|
Hbs Ag |
Non-Reactive |
- |
|
HIV Antibody |
Non-Reactive |
- |
|
Blood culture |
No growth |
- |
|
Wound culture |
MRSA growth |
- |
|
Urine culture |
No growth |
- |
|
Urine R/E |
Normal |
- |
|
Note:*normal values for female, MCV=mean corpuscular volume, PT=Prothrombin time, ALT=alanine aminotransferase, ESR=Erythrocyte Sedimentation rate, CRP=C-reactive protein. |
||
Table 1: Relevant investigations done.
Citation: Khan K and Qureshi S (2019) Overlap of Toxic Epidermal Necrolysis and Cutaneous Drug Induced Lupus-A Case Report. Emerg Infect Dis Diag J: EIDDJ-100005.