Open Access Journal of Agriculture Research
(ISSN 2651-9003)
Research Article
Biocontrol of Rhizoctoniasolani by water extracts from Chlorella sp. and Halopithys sp
Righini H1, Roberti R1* and Quintana AM2
1Department of Agriculture and Food Sciences, University of Bologna, Italy
2Spanish Algae Bank, Institute of Oceanography and Global Change, IOCAG, University of Las Palmas de
Gran Canaria, Spain
*Corresponding author: Roberta Roberti, Department of Agricultural and Food Sciences, University of Bologna, Italy, Tel: +39 051 2096568; Email: roberta.roberti@unibo.it
Citation: Righini H, Roberti R, Quintana AM (2020) Biocontrol of Rhizoctoniasolani by water extracts from Chlorella sp. and Halopithys sp. Open Acc J Agri Res: OAJAR-100029
Received date: 26 February, 2020; Accepted date: 09 March, 2020; Published date: 16 March, 2020
Abstract
Water extracts from the green microalga Chlorella sp. and from the red macroalga Halopithys sp. were evaluated for their biostimulant effects and activity against the soil-borne pathogen Rhizoctoniasolani on tomato. Extracts were applied on seed at 2.5, 5.0, 10.0 and 20.0 mg of dry biomass per mL of water.Both extracts increased seedling emergence by an average of 5.8%. Chlorella sp. extract increased seedling dry weight at all concentrations, particularly at 2.5 mg/mL (19.2%). Extract from Halopithys sp. increased dry weight only at 10.0 mg/mL (15.8%).Root rot disease caused by R. solani was reduced by Chlorella sp. extract at all concentrations, while the extract from Halopithys sp. reduced the disease at 10 mg/mL only. Both extracts did not reduce fungal colony growth; however, they caused abundant presence of hyphal cytoplasm coagulation.
Keywords: Algae; Antifungal activity; Biocontrol; Biostimulation; Soilborne pathogen; Tomato; Water extracts
Introduction
Rhizoctoniasolani J.G. Kühn (teleomorph: Thanatephoruscucumeris (A.B. Frank) Donk.) is a soil-borne fungal pathogen with a broad hostrange [1,2] such as lettuce, sugar beet, potato and tomato [3-6]. The fungus causes high crop losses in horticulture all over the world [4,7,8,9]. The control of soil borne pathogens such as R. solani is difficult, since no highly effective environmental sustainable strategies are available. The funguscan survive in the soil as mycelia or sclerotia during unfavourable environmental conditions for several years [10] and there is a lack of high resistance plants to the pathogen [11].
In agriculture, products based on algae are already used to enhance crop productivity and soil fertility [12-14] due to their content in essential nutrients, trace of metals [15] and plant growth regulators such as auxins and cytokinins [16,13].
The potential of algae against fungal phytopathogens has been highlighted in case of extracts obtained with organic solvents or alkaline hydrolysis [17-23]. Most of these studies have shown the antifungal activity of extracts from brown algae such as Ascophyllumnodosum, Eckloniasp. and Sargassum sp.A number of reports have shown that algal water extracts exert antifungal activity [24-26]. For instance, the extract from the red alga Halopithys sp. reduced zucchini powdery mildew caused by Podosphaeraxanthii [25]. About Chlorella sp., the majority of studies were focused on the plant biostimulant effects, while an antimicrobial activity was reported against human pathogens [27,28].
Considering that the European Community has restricted the placing on the market of synthetic pesticides for plant disease control and encourages alternative approaches (EC Regulation No 1107/2009; Directive 2009/128/EC), there is a need of alternatives to synthetic products. Therefore, there is an increase of interest in natural products to manage fungal plant diseases. Among these products, extracts from algae represent a source of bioactive compounds for both the bio control of fungal plant pathogen and the elicitation of plant resistance to counteract biotic stresses.
The objectives of this work was to study the effect of water extracts from the green microalga Chlorella sp. and from the red alga Halopithys sp. on (i) tomato plant biostimulation, (ii) root rot disease control on tomato plants grown in substrate infected with R. solani and (iii) R. solanigrowth.
Materials and Methods
Water extracts preparation, plant material and pathogen: Chlorella sp. and Halopithys sp. were gently provided by the Spanish Bank of Algae (BEA), University of Las Palmas de Gran Canaria. Lyophilized biomass of Chlorella sp. and grinded dry thallus of Halopithys sp. were suspended in sterile distilled water (0.5%) under continuous stirring at 50 °C for 12 h and thenfiltered [29]. For the pathogenicity test and the in vivo experiments, tomato seeds cv. “Perad’Abruzzo” (Blumen Group S.p.A., Milano, Italy) were used. The fungus Rhizoctoniasolani 3001 belonging to DISTAL collectionwas isolated from tomato plant tissue showing symptoms of root and crown rot. Pieces of symptomatic tissues were surface disinfected with 2.0% sodium hypochlorite solution for 2 min, rinsed three times in sterile distilled water and placedon potato dextrose agar (PDA 3.9%, BiolifeS.r.l., MI, Italy) added with 60 mg l/L of streptomycin sulphate (Sigma - Aldrich Co.). After 7days in the dark at 25 °C, the presence of R. solani from tomato tissues was examined using a light microscope (Carl Zeiss mod. ZM, Germany) at × 500 magnification. The fungus pathogenicity was verified through inoculation of 7-day-old colony portions on tomato root seedlings and waiting for the symptom appearance.
Effect of seed treatment with water extracts on seed emergence and seedling dry weight: Tomato seeds were sterilized following [30] method with modifications. Seeds were surface-disinfected in 2.0% sodium hypochlorite solution for three minutes and then rinsed three times in sterile distilled water. Seeds were treated by immersion in 1-mL aliquot of each concentration extract at 2.5, 5.0, 10.0 and 20.0 mg/mL of Chlorella sp. and Halopithys sp. overnight in the dark. Seeds immersed insterile distilled water were used as control. After treatment, seeds were sown in a substrate consisting of a mixture of peat and sand (7:3, w:w) in plastic trays. Thirty seeds per tray were sown and four trays (replicates) for each concentration and the control were considered. Trays were arranged in a completely randomized design on a shelf of a growth chamber at 24-26 °C (day), 20-22 °C (night), 14 h photoperiod, 70% relative humidity. Two weeks after sowing, seedling emergence was recorded. Three weeks after sowing seedlings were gently removed from the substrate, washed with tap water and dried in a hoven at 60 ± 5 °C for 72 h, and then seedling dry weight was determined. The experiment was repeated twice.
Effect of seed treatment with water extracts against R. solani: Tomato seeds were treated with water extracts as reported above and then sown in the substrate previously inoculated with R.solani. For substrate inoculation, 10-day-old colonies of the pathogen grown on PDA medium were blended with sterile distilled water and mixed with the substrate (3.0% w:w, pathogen: substrate). Seeds immersed in sterile distilled water were used as control. Four trays (replicates) were considered for each concentration and for the control. Trays were incubated in the growth chamber at the same conditions reported above. Four weeks after sowing, seedlings were removed, washed with tap water and necrosis root symptoms caused by R. solani were visually assessed. The severity of disease was determined by evaluating root rot on a scale of 0 to 100, where: 0=no symptoms; 5=slight necrosis; 20=moderate necrosis; 50=severe necrosis; 100=very severe necrosis and dead plant [31], modified. For dry weight determination, the infected seedlings were dried in a hoven at 60 ± 5 °C for 96 h. The experiment was repeated twice.
Effect of water extracts on R. solani growth: Fungal portions (7 mm diameter) were cut from 10-day-old colony then treated by immersion in 1-mL aliquot of each extract concentration. Portions immersed in sterile distilled water were used as control. Six hours after treatment, colony portions were placed on the surface of PDA medium in Petri dish. Four dishes (replicates) were used for each concentration and for the control. The dishes were incubated at 24-25 °C in the dark for 7 days. Colony diameters were measured daily along two perpendicular axes. Hyphal cytoplasm coagulation of the same colonies was evaluated on four portions (replicates) by using an Eclipse TE2000-E microscope (Nikon Instruments Europe BV, Amsterdam, The Netherlands) at × 600 magnification. Hyphal cytoplasm coagulation was assessed by using the following scale: 1, 0-10% of coagulation; 2, 11-40% of coagulation; 3, 41-80% and 4, >80% of coagulation. The experiment was repeated twice.
Statistical analysis: All experiments were conducted by using completely randomized design. Data were first examined for distribution normal before analysis of variance (ANOVA), and percentage values were arcsine transformed before ANOVA. Two-way ANOVA was applied to test the main effects and interaction of algal water extracts and concentrations against all the parameters. LSD Multiple Range Test was used to separate the means. The software Statgraphics Plus 2.1, and statistical significance at P < 0.05 was used.
Results
The effect of tomato seed treatment with different concentration of the extracts from Chlorella sp. and Halopithys sp. on seedlings is showed in Figure 1. Two-way ANOVA analysis indicated that for the emergence only the concentration factor is significant. All concentrations significantly increased the emergence with similar values (5.8%) with respect to the control (0.0 mg/mL).
Concentration factor is significant according to two way ANOVA. F (4, 100) = 3.16, P< 0.05. Columns are mean values ± SD. Different letters indicate significant differences, according to LSD test (P< 0.05).
For seedling dry weight, significant interaction was found between extract and concentration factors (Figure 2). Dry weight was significantly increased by extract from Chlorella sp. at 2.5, 5.0, 10.0 and 20.0 mg/mL by 19.2, 10.4, 14.2 and 12.7%, respectively and from Halopithys sp. at 10.0 mg/mL by 15.8%. Concerning the comparison of concentrations, the highest values of dry weight were obtained with 2.5, 5.0 and 20.0 mg/mL of Chlorella sp.
Extract and concentration factors and their interaction are significant according to two way ANOVA. F (1, 40) = 46.87, P< 0.05 (for alga factor), F (4, 40) = 20.25, P< 0.05 (for concentration factor), F (4, 40) =9.66, P< 0.05 (for interaction). Columns are mean values ± SD. The asterisk indicates significant dry weight increase by each concentration towards the corresponding control (0.0 mg/mL) and different letters indicate significant differences within each concentration, according to LSD test (P< 0.05).
The effect of tomato seed treatment with the extracts against R. solaniis showed in Figure 3. Two-way ANOVA of disease severity revealed that extract and concentration factors and their interaction were significant. The treatment with Chlorella sp. was the most effective. All concentrations of Chlorella sp. extract reduced the disease by 54.9, 48.2, 54.9 and 35% at 2.5, 5.0, 10.0 and 20.0mg/mL, respectively, towards to the untreated control. Halopithys extract significantly reduced the disease severity only at 10.0 mg/mL by 18.0%. Within effective concentrations, 10 mg/mL of Chlorella sp. reduced the disease more than Halopithys sp.
Extract and concentration factors and their interaction are significant according to two way ANOVA. F (1, 40) = 279.67, P< 0.05 (for alga factor), F (4, 40) = 37.86, P< 0.05 (for concentration factor), F (4, 40) = 21.09, P< 0.05 (for interaction). Columns are mean values ± SD. The asterisk indicates significant reduction by each concentration towards the corresponding control (0.0 mg/mL) and different letters indicate significant differences within each concentration, according to LSD test (P< 0.05).Disease severity was calculated on a scale of 0 to 100.
Figure 4 shows the effect of the extracts on dry weight of infected seedlings. Two-way ANOVA indicates that dry weight was influenced by algal extract, concentration and their interaction. The extract from Chlorella sp. was the most effective extract in increasing dry weight. It was increased by all extract concentrations by 24.0, 11.6, 12.4 and 6.8% at 2.5, 5.0, 10.0 and 20.0 mg/mL, respectively, with respect to the control (0.0 mg/mL). Extracts from Halopithys sp. increased dry weight only at 10 mg/mL by 17.1%.
Extract and concentration factors and their interaction are significant according to two way ANOVA. F (1, 40) = 60.35, P< 0.05 (for alga factor), F (4, 40) = 30.87, P< 0.05 (for concentration factor), F (4, 40) = 22.10, P< 0.05 (for interaction). Columns are mean values ± SD. The asterisk indicates significant dry weight increase by each concentration towards the corresponding control (0.0 mg/mL) and different letters indicate significant differences within each concentration, according to LSD test (P< 0.05).
In Table 1 the effect of the extracts from Chlorella sp. and Halopithys spon R. solaniis shown. Fungal colony growth was not affected by the extracts at any concentration. As regards cytoplasm coagulation, two-way ANOVA indicates a significant interaction between extract and concentration factors. Both Chlorella sp. and Halopithys sp. extracts at all concentrations increased cytoplasm coagulation with respect to the control (0.0 mg/mL). Extract from Chlorella sp. increased cytoplasm coagulation by an average of 53.8%, and that from Halopithys sp. by 80.4, 63.0, 202.2 and 178.3% at 2.5, 5.0, 10.0 and 20.0 mg/mL, respectively. At 10.0 and 20.0 mg/mL, Halopithys sp. extract showed the highest increase of cytoplasm coagulation.
For colony growth, two way ANOVA was not significant. For cytoplasm coagulation, extract and concentration factors and their interaction are significant according to two way ANOVA. F (1, 40) = 51.29, P< 0.05 (for alga factor), F (4, 40)=33.81, P< 0.05 (for concentration factor), F (4, 40) = 12.70, P< 0.05 (for interaction).Means ( ± SD) followed by different lower-case letters in a column and by different upper-case letters in a line are significantly different according to LSD test (P< 0.05). The absence of lower- or upper-case letters indicates no significantly differences.
Discussion
Algae and cyano bacteria extracts are commonly used on several horticultural crops for their capacity to improve plant growth and yield [12-14,32]. The antifungal activity of the extracts obtained with organic solvents has been widely investigated [17-21] while few studies have examined the activity of water extracts [25,26,29].
In this work, we present data of a first study on the effect of water extracts from the green microalga Chlorella sp. and from the red macroalga Halopithys sp. on tomato, both as biostimulant and against the soil-borne pathogen Rhizoctoniasolani. The experiments showed that both biostimulant and antifungal effects depend on the algal extract and its concentration. This is in accordance with what reviewed by [33,34] and observed by [35] on rocket and by [36] on wheat. Our experiments also showed that tomato emergence was enhanced by seed treatment at all extract concentrations. Moreover, extract from Chlorella sp. increased seedling dry weight in line with what reported by [36] on wheat [37] also observed an increase of fresh weight of Chinese chives and spinach after treatment with Chlorella fusca. A biostimulant effect on tomato was obtained by [38] with an extract from the green microalga Acutodesmusdimorphus applied as seed and foliar treatment. These treatments enhanced seed germination, plant height, numbers of flowers and branches per plant in a dose dependent manner. Indeed, green microalgae contain high levels of micro and macronutrients essential for plant growth. As reported by [39], extracts from microalgae are characterized by high content of carbohydrates and proteins that can reach 55-70 % of their fresh weight. Indeed, carbohydrates and proteins can constitute up to 46% and 18-46% of their dry weight extract, respectively [40-43]. The biostimulant behaviour of microalgae can be related to the presence in their extracts of substances, such as the amino acids tryptophan and arginine, precursors of phytohormones such as auxin anssalycilic acid [44,45].
Concerning the effect of Chlorella sp. and Halopithys sp. against R. solani, the extracts from Chlorella sp. was the most active in reducing root rot disease. To our knowledge, only [46] reported a biocontrol activity of Chlorellavulgaris, mixed with others microorganisms, against Botrytiscinerea on strawberry fruits both under field conditionsand after storage. This extract was also able to inhibit the B. cinerea mycelial growth and sporulation in in vitro experiments, in contrast with our results. Water extracts from Halopithys sp. reduced R. solaniroot rot only at 10 mg/mL, while in our previous study, 5.0 mg/mL of the same extract reduced powdery mildew disease and pathogen sporulation on detached zucchini cotyledons [25]. The control of R. solani disease can be related to the presence of carbohydrates such as carrageenans the major components of the extracellular matrix in red algae [47,48]. Carbohydrates are also elicitors of plant defence responses against pathogens as demonstrated for the extract from the red alga Kappaphycusalvareziion tomato seedlings, where the treatment increased transcription of pathogenesis related genes such as PR-1b1, PR-3 and PR-4 with and without M. phaseolinachallenge [49].
We underline that water extracts from Chlorella sp. and Halopithys sp. caused an increase of R. solani hyphal cytoplasm coagulation even if they did not reduce the colony growth. We consider this morphological alteration an antifungal activity, because it is indicative of unfavourable environmental conditions that could be related to bioactive compounds in the water extracts. Fungal morphological alterations were observed in Aspergillus niger after treatment with the essential oil of Cymbopogonnardus that induced cytoplasmic granular aspect with vesicular structures and cell wall disruption [50]. On B. cinerea, treatment with the phenol pterostilbene caused big vacuoles, hyphae swollening and high presence of vesicles [51]. It is known that algae are a source of natural products with a broad spectrum of biological activities such as antimicrobial [27], antifungal [26] and antioxidant activities [52]. As reported by [53], the antimicrobial activity of green and red algae is related to the presence of polysaccharides, fatty acids, phlorotannins, pigments, lectins, alkaloids, terpenoids and halogenated compounds. Another study correlates the antifungal activity of green microalgae such as Zygenmaczundae, Zygenmastellinum and Zygenma tenue against Curvularialunata, Fusariumsporotrichoides, M. phaseolina, R. solani and Sclerotiumrolfsii with their content in fatty acids, sterols and terpenes [54].
Conclusion
In conclusion, this study shows that tomato seed treatment with water extracts from the green microalga Chlorella sp. and from the red macroalga Halopithys sp. induced plant biostimulation and have potential to control R. solani root rot.
Considering that the control of the pathogen is currently problematic for the limited available pesticides, these extract may be considered a useful tool to be exploited for the disease management in sustainable agriculture, once their effectiveness will be verified on tomato plants in a larger scale experiment.
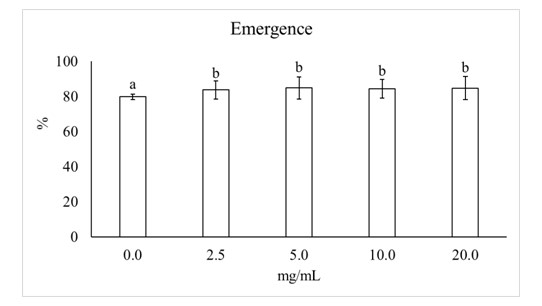
Figure 1: Effect of seed treatment with different concentrations of water extracts from Chlorella sp. and Halopithys sp. on tomato seedling emergence.
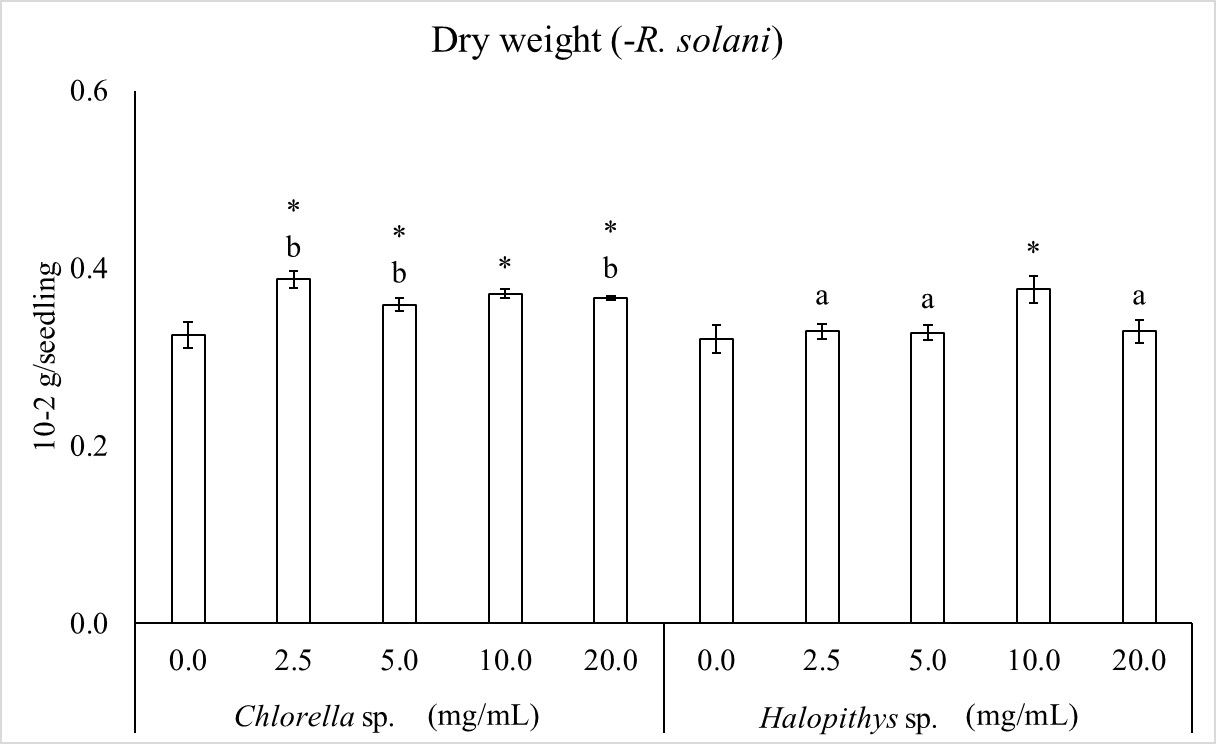
Figure 2: Effect of seed treatment with different concentrations of water extracts from Chlorella sp. and Halopithys sp. on tomato seedling dry weight.
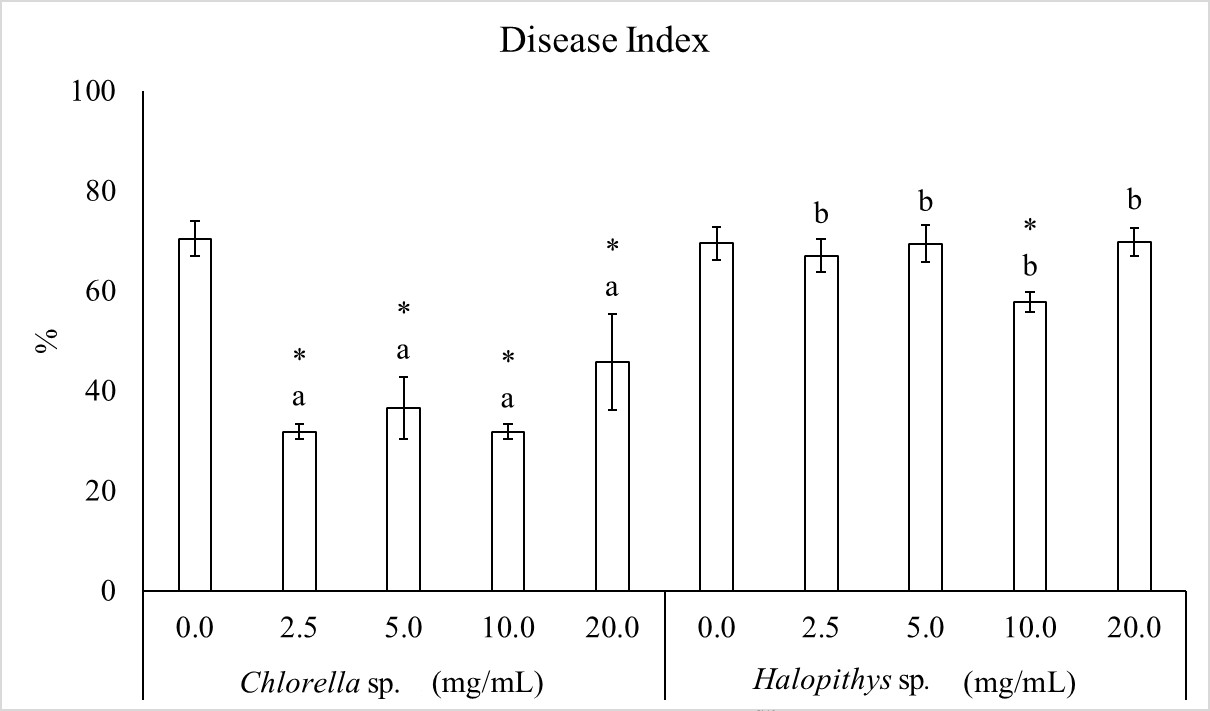
Figure 3: Effect of tomato seed treatment with different concentrations of water extracts from Chlorella sp. and Halopithys sp. on seedling root rot caused by R. solani.
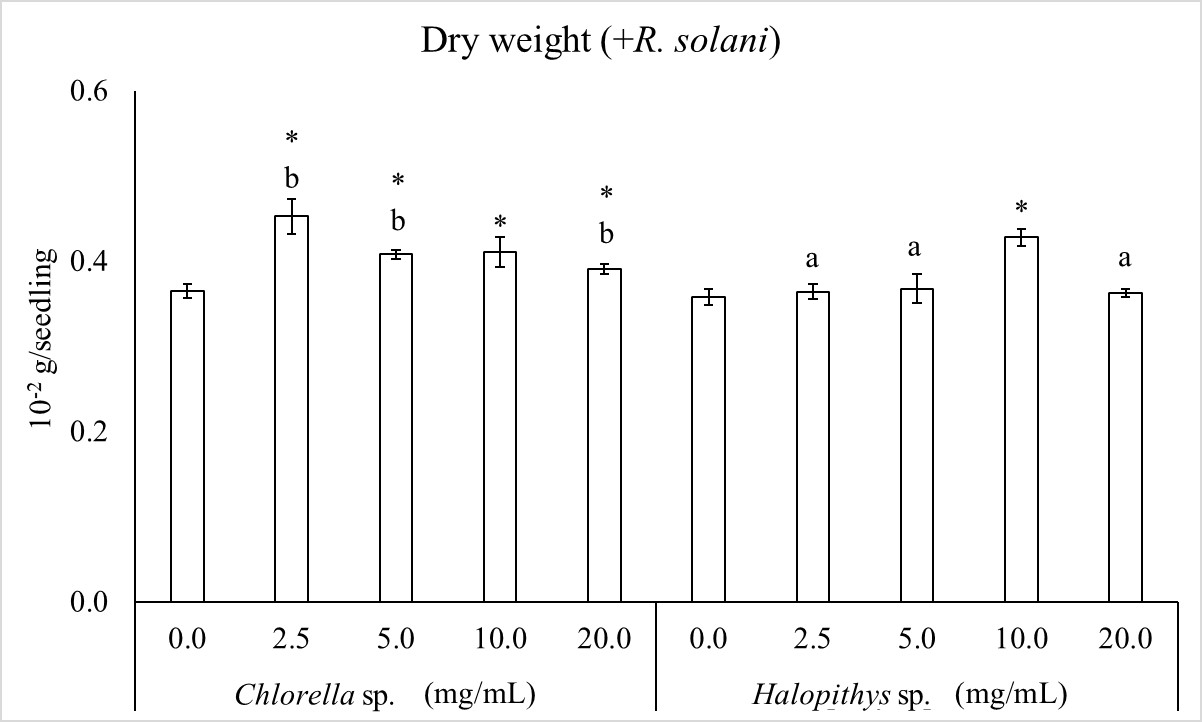
Figure 4: Effect of tomato seed treatment with different concentrations of water extracts from Chlorella sp. and Halopithys sp. on dry weight of seedlings infected by R. solani.
|
Extract |
mg/mL |
||||
|
0 |
2.5 |
5 |
10 |
20 |
|
|
Colony growth (mm) |
|||||
|
Chlorella sp. |
33.0 ± 4.5 |
34.6 ± 2.6 |
33.0 ± 4.5 |
32.1 ± 1.3 |
34.7 ±1.2 |
|
Halopithys sp. |
32.0 ± 4.7 |
34.9 ± 1.8 |
33.3 ± 2.8 |
33.5 ± 1.9 |
33.9 ± 1.7 |
|
Cytoplasm coagulation |
|||||
|
Chlorella sp. |
1.2 ± 0.2 A |
1.8 ± 0.2 B |
1.6 ± 0.3 B |
1.9 ± 0.3 Ba |
1.8 ± 0.3 Ba |
|
Halopithys sp. |
1.1 ± 0.1 A |
2.1 ± 0.2 C |
1.9 ± 0.3 B |
3.5 ± 0.4 Bb |
3.2 ± 0.5 Cb |
Table 1: Effect of treatment with different concentrations of water extracts from Chlorella sp. and Halopithys sp. on Rhizoctoniasolani.
Citation: Righini H, Roberti R, Quintana AM (2020) Biocontrol of Rhizoctoniasolani by water extracts from Chlorella sp. and Halopithys sp. Open Acc J Agri Res: OAJAR-100029