Current Trends in Biotechnology and Biochemistry
Research Article
Two-Way Roles of Fibronectin Binding Proteins in Cell Adhesion and Cross-Dressing in Mycobacterium Tuberculosis H37Rv Pathogenesis
Meena S and Meena LS*
CSIR-Institute of Genomics and Integrative Biology, India
*Corresponding author: Laxman S. Meena, CSIR-Institute of Genomics and Integrative Biology, India, Tel: 011-27666156; Email: meena@igib.res.in
Citation: Meena S and Meena LS (2019) Prescience of Rv1786 of Mycobacterium tuberculosis H37Rv to assist the pathogenic potentials and significance by computational annotations. Curr Trends Biotechnol Biochem: CTBB-100007
Received date: 21 August 2019; Accepted date: 27 August, 2019; Published date: 02 September, 2019
Abstract
Mycobacterium tuberculosis is a pathogen responsible for worldwide pandemic Tuberculosis. Host invasion requires multiple adhesion factors in this pathogen. Fibronectin binding proteins is a class of adhesion protein, which belongs to PE_PGRS group of proteins in M. tuberculosis. FnBPs help in adhesion and exhibit binding with extracellular surface receptor Fibronectin via transmembrane integrin receptor of the host cell. Despite of general presentation of antigen, bacilli deploy a strategic mechanism to cross-dress other uninfected immune cells. Cross-dressing is a mode of antigen presentation which can be described as the transfer of preformed MHC-II-antigen complex to the surface of recipient cell. Further, these cross-dressed cells sooner or later present the intact or unprocessed MHC-antigen complex to other lymphocytic cells, which helps mycobacterium to evade the induced immune response. In this review, we discuss the recent research on cross-dressing phenomenon, which is primarily carried out via exocytosis, trogocytosis and tunneling nanotube formation. Calcium is established to play a major role in the formation of membranous structures and thus might govern the cross-dressing phenomenon. Therefore, targeting this central pathway of transfer of FnBPs to uninfected immune cells can help in devising a novel therapeutic option for TB treatment.
Keywords: Fibronectin; Fibronectin binding protein; M. tuberculosis; PE_PGRS; T-cells
Abbreviations
DC : Dendritic cell
FnBPs : Fibronectin binding proteins
MHC : Major histocompatibility complex
M. tuberculosis : Mycobacterium tuberculosis
MVB : Multi vesicular bodies
MVs : Microvesicles
PE-PGRS : Proline Glutamic polymorphic GC rich repetitive sequence
TNT : Tunneling nanotubes
TB : Tuberculosis
Introduction
Tuberculosis (TB) is a deadly disease, which critically affects public health worldwide. The causative agent of TB is a gram positive bacterium, Mycobacterium tuberculosis (M. tuberculosis). TB, remains a formidable infectious disease with global signi?cance, around 75% of the world’s population is infected with TB the World Health Organization (WHO) Global Tuberculosis Report indicated that in 2017 the best estimate is that 10.0 million individuals (range, 9.0-11.1 million) developed Tuberculosis disease: 5.8 million men, 3.2 million ladies, and 1.0 million kids. Drug-resistant Tuberculosis continues to be a public health crisis. The most effective estimate is that, worldwide in 2017, 558 000 people (range, 483 000-639 000) developed Tuberculosis that was resistant to rifampicin (RR-TB), the foremost effective first-line drug, and of these, 82 had multidrug-resistant Tuberculosis (MDR-TB). 3 countries accounted for pretty much half of the world’s cases of MDR/RR-TB: India (24%), China (13%) and therefore the Russian federation (10%) [1]. The reasons for successful survival of M. tuberculosis within host cell are still not well known. Bacilli release mycobacterial components from the phagosome during infection, which hampers with intracellular membrane trafficking in infected macrophages [2]. Secretion of virulence factors within or outside the infected host cell further facilitates the survival abilities of M. tuberculosis. Eukaryotic cell channelizes the secretion and absorption of varied micro and macromolecule via membrane trafficking. Most of the cell secretions are secreted through membrane derived extracellular vesicles like exosomes and micro vesicles (MVs) [3]. Immune cells require intercellular communication for their crucial functions like migration and signaling, to elicit an appropriate immune response. Cross dressing is presentation of preformed MHC antigen complex by dendritic cells in other hand during cross presentation antigen is acquired by dendritic cell than processed and present the antigenic peptide on them owns MHC protein’s [4]. In general, presence of bacilli in the host macrophage produces a variety of cytokines, which is responsible for the onset of innate immune system [5-7]. Apart from well-studied soluble factors of intercellular communication, physical factors like exosomes, tunneling nanotube (TNTs) and direct transfer in case of trogocytosis has been recently studied [7]. These processes are used by M. tuberculosis virulence factors which play an imperative role to modulate the host immune system through subsequent cross-dressing of various immune cells [8]. These membranes derived physical structures are gaining interest as they are involved in the presentation of antigen and thus represent a new strategy employed by M. tuberculosis for enhanced survival. Recent studies have shown that exosomes carry genetic materials like mRNA as well as virulence factors of M. tuberculosis. Generation of exosomes is an alternate approach of transferring important messages from cell to cell [9]. Few biomarkers related to exosomes, TNTs, trogocytosis have been researched out and are proposed to be landmarks for the diagnosis of TB cases [10]. Hence, M. tuberculosis is one of the most successful intracellular pathogens, which can resist a variety of defense mechanisms, and alters the immunological events of host macrophages during infection [11].
Fibronectin Binding Proteins (FnBPs)
M. tuberculosis has various antigens by which it can modulate the host immune system [12]. FnBPs are confined to the outer surface layer of M. tuberculosis and they are the primary secretory antigens responsible for pathogen’s adherence and invasion in initial phases of TB establishment. It is of 30-31 kDa protein which is found prominently in short term culture soup of M. tuberculosis [13]. Exosomes, TNTs and trogocytosis help in cross-dressing of mycobacterium antigenic factors to the immune cells [14]. In M. tuberculosis several FnBPs are studied like Antigen 85 complex (Ag85), which have three different proteins 85A, 85B and 85C which encoded by three different genes Rv3804, Rv1886 and Rv0129 respectively [15,16]. In a study it has been proved that Ag85 complex involved in adherence and invasion in to the host as they show the activity of Fn binding at a particular motif which is confirmed by peptide inhibition assay [17]. Mesenchymal stromal cells (MSC) show anti-inflammatory response [18]. MSC infusion induced immune refocusing of M. tuberculosis antigen-directed T-cell responses, resulting in improved recognition of Ag85B [19]. Additional family of FnBPs belongs to PE_PGRS and has approximately 60 genes. Some of PE_PGRS proteins exhibit FnBPs activity, which binds to the host fibronectin via integrin receptor. For examples, Rv3652 and Rv3653 encode PE_PGRS60 and PE_PGRS61 respectively, in M. tuberculosis. ELISA quantitation of Ag85B in lung homogenates of M. tuberculosis infected mice exposed that although Ag85B gathers in the lungs as the bacterial population expands, the amount of Ag85B per bacterium decreases nearly 10,000-fold at later stages of infection, coincident with the growth of T cell responses and arrest of bacterial population growth. These results show that bacterial protein secretion in vivo is active and controlled [20]. Both the proteins are surface associated and exhibit FnBPs binding properties as well [21,22]. It was also demonstrated that Rv1759c gene of M. tuberculosis which is a member of PGRS family [24] and it is also a close relative of PE_PGRS81. It expresses an adherence protein which is reported as Fn binding protein as it binds with bacterial surface coated with Fn [25,26]. This manuscript shows the knowledge, whether FnBPs of M. tuberculosis expressed by the PE_PGRS protein family member is cross dressed by exosomes, TNT or trogocytosis to alter the immune system of the host in TB infection (Figure 1).
Importance of Cross-Dressing in M. Tuberculosis Pathogenesis
Exosomes involvement: Most of the eukaryotic cells continuously secrete small membrane bound vesicles i.e. exosome, ranging 30-100nm into lymphatic and blood system to support intra and inter cellular connections. It is suggested that these vesicles are generate due to invagination of the endosomal membrane to form the internal vesicles of MVBs. There are noticeably two main functions of MVBs, first is to facilitate degradation by merging with lysosomes and secondly they integrate with plasma membrane and secrete their inner small vesicles as in form of exosome into extracellular milieu, those contain various components. This clarifies the presence of variety of cytosolic proteins, transmembrane proteins, receptors and other antigenic molecules with their exposed binding sites [7]. Due to its lumen components, exosomes are ideal and innovative sources of biomarkers in various prolonged diseases. Exosomal constituents seem to be encouraged by inflammation and hypoxic condition, which is directly related to the origin of cell, cellular anomalies and state of disease. We put forward that largely mycobacterial proteins which are present in serum are kept in exosomes and these exosomes releases continuously. Variety of virulent proteins of mycobacteria such as HspX, DnaK, and antigen 85complex are carried by exosomes. It is still unidentified that a single exosomes contain processed and unprocessed antigen or both. There is also a possibility that an antigen form will alter the cell as well as cellular reaction which is directed via exosomes constituents [22]. Exosomes basically part of intercellular trafficking as they work as a porter. Released exosome of infected macrophage and other immune cells contain significant amount of antigens which is efficiently cross activate other immune cells to generate huge amount of immune response [27]. Sometimes exosomes also named as a Trojan exosome, as they can be targeted by retrovirus to take benefit of the intracellular vesicular trafficking and exosomal interchange pathway to move among cells in search of ample target cells [28]. Exosomes also carry miRNA which involved in regulation of immunity, including lymphocyte development and differentiation and cytokine production. Recent finding shows that T CD8+ cell derived suppressive exosomes shipping miRNA-150, that behave antigen-specifically, to inhibit murine contact sensitivity response indirectly by affecting antigen presenting cells, especially macrophage [29]. Another research proposes that some of miRNA like miR-155 and miR-146a are released through exosome from DC, which critically control the inflammatory responses [14]. miRNA expression can be used as biomarker for detection of TB. Expression of miR-484, miR-425 and miR-96 were signi?cantly amplified in serum of TB patients which interrelated with the TB infection level [30]. Earlier research reveals that some of the tumors cells also released exosomes, those carry tumor specific antigen, and induce antigen specific immunity against autologous tumors. Exosomes carry antigen and adjuvant properties which provide the capability to induce strong immune response [29] (Figure 2). Future studies will focus on deciphering the role of FnBPs in cross dressing and how this phenomenon can govern M. tuberculosis pathogenesis. So this leads a path on which we can use exosome as a novel family of vaccines.
TNT involvement: Now another aspect of antigen presentation is tunneling nanotubes. TNTs are slender membranous actin structures which linking distant cells and allow the transfer of wide array of cellular signaling molecule and various other things like vesicles, organelle (mitochondria) [30], proteins and ions (Ca2+). These structures earlier shown in rat cultured pheochromocytoma PC12 cells (cells of adrenal medulla that have an origin from the neural crest and mixture of neuroblastic and eosinophilic cells) then subsequently found in various cells and almost all immune cells invivo and invitro [7]. Some of the pathogenic antigen might be involve in promoting TNTs formation [31]. Recently it is revealed that in neuronal disease, prion proteins takeover the TNTs, to extend intracellularly in other uninfected neurons of central nervous system [32]. TNTs formed in different cells shows different structural and functional differences. TNTs, formed in infected macrophages are permeable for Ca2+ in contrast to TNTs formed in T-cells and also have connections that are anatomically dissimilar to the open ended membranous tethers present in various cells together with macrophages. Recently, it is highlighted that viruses like, HTLV-1 and HIV-1 have been presumed to use TNTs to travel intercellular [33]. TNTs provide more efficient way for microorganisms to spread as it decreases the requirement to enter extracellular milieu which minimizes their exposure to various anti-pathogenic factors [32]. In recent research nanotubes formation enhance by IgE Immunoglobulin antigen receptor and chemokine receptor-1 (CCR1) in mast cells. Cells those lacking CCR1 receptor on their cell surface fails to trigger the formation of nanotubes. Experimentally nanotubes formation enhances by using nanostructures like Haptens (like 2, 4-DNP) and impacting the cellular signaling cascade. The extent and occurrence of these membranous nanotubes seem to fluctuate according to spatial circulation of hapten nanostructures. So, nanotechnology found to be novel way to study a chosen cellular signaling by activating them [34]. In latest study using Bacillus subtilis (B. subtilis), nanotubes are chain of fragments of membranous chain built continuous lumen. Nano-tubes are formed rapidly and where cell concentration is low, it is found in intercellular and elongated tubes. It has been said that YmdB (Uncharacterized protein thought to involve in distability in bacillus) which is calcinuerin like protein, essential to develop nanotube and also participates in intercellular trading. YmdB is a phosphodiesterase which is evidenced to down regulate execution of motility genes and enhance the genes involved in bio-film development [35-37]. Nano-tubes are also formed in inter-specific manner as they provide a path to intercellular trafficking within and across species. Nano-tubes could also elongate and may give bud to free MVs which depend on environmental and physiological conditions [38-39]. In respect of M. tuberculosis due to its macrophage resident nature it might also infect another healthy macrophage and its antigens transfer through TNTs like FnBPs and transiently activate other immune cells to enhance immune response (Figure 2). This needs lighter to lead a way to know the other aspects of pathogenicity of M. tuberculosis.
Trogocytosis involvement: Now the third aspect of antigen presentation is trogocytosis in which exchange of membrane fragments occur. It is well known that cell surface protein considered specific to particular cell but later on through some process they can be detected on other cell types [40]. Trogocytosis is a phenomenon through which temporary contact dependent uptake of membrane takes place [41]. It is actin based motility structure through which various bacterial antigen transfer intercellular. Macrophages are also able to interact with adjacent cells through trogocytosis. Transfer of antigens surrounded by two layers of membrane, one of each donor and acceptor cell occurs [42,43]. After that, cells would eventually separate and retention of the transferred membrane occurs till they are recycled by new membrane fractions. Trogocytosis improves the impact of infection and immunity in immune cells via improvising T-cell signaling in response to antigen acquired by dendritic cells from neighboring cells [40]. In a recent study it has been shown that rate of trogocytosis increased during primary cell infection. This suggest that some kind of triggering signaling molecule which implicate in infected cell to initiate, enhanced and stabilize the trogocytosis process [42] (Figure 2). In aspect of M. tuberculosis, increasing rate of infection this process might play an important role to invade new macrophage cells. Its various pathogenic antigen specially FnBPs, which primarily involve in adhesion and invasion of cells use this new way to cross dressed other immune cells and enhance immune response.
Ca2+ involvement during cross-dressing: Role of Ca2+ is indispensable in membrane fusion through various signaling pathway. During early stage of infection bacilli suppresses the cytosolic Ca2+ concentration, which inhibits the fusion of phagosome-lysosome [44]. Ca2+ plays a crucial role in various phenomenon related to cross-dressing. Cross-dressing is done by membranous structure to present the antigens on the surface of the cell membrane. According to one of the research, it is shown that if cells are treated with phorbol ester or Ca2+ ionophore there is increase in secretion of exosomes. In contrast, if cells are treated with phosphatidyl inositol 3-kinase inhibitor and wortmannin, secretion of exosomes decreases [45]. In previous study, release of exosome was markedly enhanced due to treatment of monensin (MON), which is Na+/H+ exchanger that increase intracellular Ca2+ concentration [46]. It needs a handful research to know better aspects of Ca2+ signaling in cross dressing. M. tuberculosis effect on host cells are channelized through change in various signaling machinery [47]. So, bacilli interfere with the PI3K activation, which is an important for cell survival and growth. M. tuberculosis also affects its downstream effectors such as Ca2+ Calmodulin pathway [48]. Ca2+ signaling is one of the most extensively researched pathways in eukaryotes and it is also foremost effector molecule of stimulus responses which is associated to various physiological processes. Ca2+ binding protein like calmodulins are involved in transduction of Ca2+mediated responses [49] (Figure 1). In prokaryotes Ca2+ signaling are less studied. Calmodulin like protein (CAMLP) in prokaryotes was first shown in B. subtilis and E. coli [50, 51]. Soon CAMLP is also found in various strains of mycobacterium [52-56]. The CAMLP coding genes are Rv1211 and Rv1212c in M. tuberculosis. As in earlier studies it has shown that, CAMLP induce the action of bovine brain, Phosphodiesterase and plant kinases, which elucidates eukaryotic target of calmodulin [57]. The gene Rv1211 is TraSH, needs Ca2+ during development and persistence of M. tuberculosis in host cell [58]. Rv1211 encodes a protein with calmodulin-like activity (CAMLP) in the human pathogen M. tuberculosis and acts as a potential target for trifluoperazine [58]. So Ca2+ involvement during cross-dressing is indispensable to enhance immune response of host.
Discussion
A strange dispersal of various adhesive and invasive proteins associated to the membrane of M. tuberculosis and how they participate in host pathogen interaction remains vague. This review provides a mode of antigen presentation through cross-dressing by variety of methods. Specifically, an emphasis on adhesive antigen FnBPs, which belong to PE_PGRS group of family proteins of M. tuberculosis is given. It is well understood that FnBPs binds to Fn and provide adhesion between host and pathogen which is a first and foremost step in infection. M. tuberculosis uses cross-dressing as a protective mechanism by which it can easily pass out the immune response and effectively enter into latent phase of disease. Nowadays this mechanism of antigen presentation is gaining interest as it provides a new mode of pathogenicity to alter host immune response. In these mechanisms Ca2+ is required as they involved the formations of membranous fragments. As Ca2+ is the main downstream effectors molecule of PI3K pathway, which is an essential signaling cascade of M. tuberculosis pathogenesis. In response of Ca2+ signaling, prokaryotic Ca2+ binding proteins such as CAMLP, take over the charge to initiate various signaling cascade. In this way, we can say that change in Ca2+concentration affect the formation of variety of membranous structures involved in cross-dressing. This field requires extensive research to evolve various pathways involved in pathogenesis mechanism, so that many options will be opened to create new drugs.
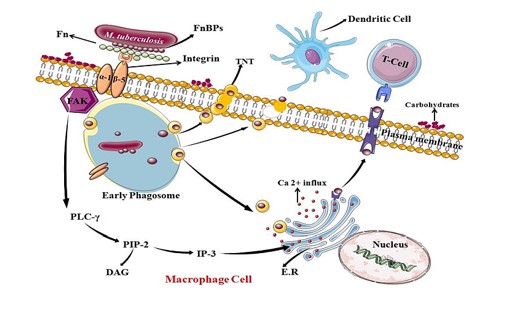
Figure 1: Graphic representation of FnBPs adhesion in macrophage cell during M. tuberculosis pathogenesis: FnBPs in M. tuberculosis which belongs to PE_PGRS protein family play role in extracellular adhesion to fibronectin through integrin receptor; here alpha (α) and beta (β) subunit serve as a docking site. This interaction facilitates tyrosine phosphorylation of Focal adhesion kinase (FAK) which in turn initiates a signaling cascade of PLCγ. It shows catabolic activity on its substrate PIP3 which release IP3 and DAG. IP3 works on endoplasmic reticulum (ER) directing to open Ca2+ channels, thus rise in Ca2+ concentration in host cell change in the actin arrangement which provide path to mycobacterium to invade the cell.
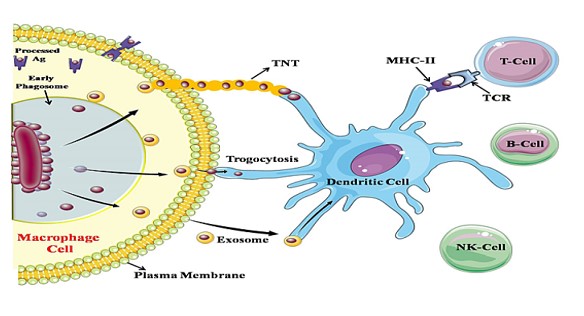
Figure 2: Schematic representation of cross dressing of Antigen to uninfected cells: Due to increase in Ca2+ concentrations various exosomes containing FnBPs are released through phagosomes. Some of the exosomes are collect outside of the plasma membrane and form nanotubes which expressed FnBPs to dendritic cell. Due to actin deformation one of the exosomal membranes diffuse to plasma membrane and antigen is directly presented by trogocytosis. Some exosome evolved out through macrophage which engulf by other dendritic cell or other macrophage, now this crossly present antigen is processed and offer to other cells with MHC-II.
Citation: Meena S and Meena LS (2019) Prescience of Rv1786 of Mycobacterium tuberculosis H37Rv to assist the pathogenic potentials and significance by computational annotations. Curr Trends Biotechnol Biochem: CTBB-100007